Contact Us
Have a question? If you can’t find what you’re looking for, contact our customer service team via contact us page, or call 09 379 9778.
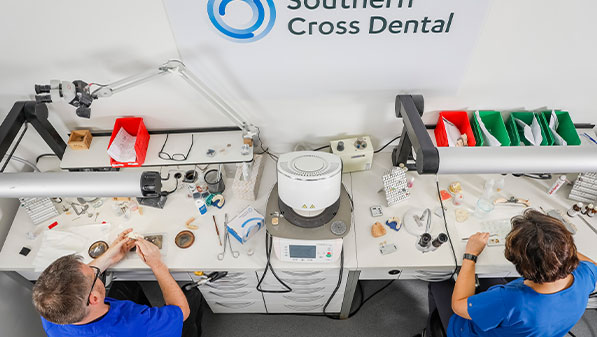
In New Zealand, most dental laboratory products are categorised as a ‘custom-made medical device’ for regulatory purposes.
We adhere to the highest levels of quality management to ensure that our products are made in compliance with all government and Medsafe regulations. The safety and reliability of our products sets the standard for high-quality dental products.
Contact Us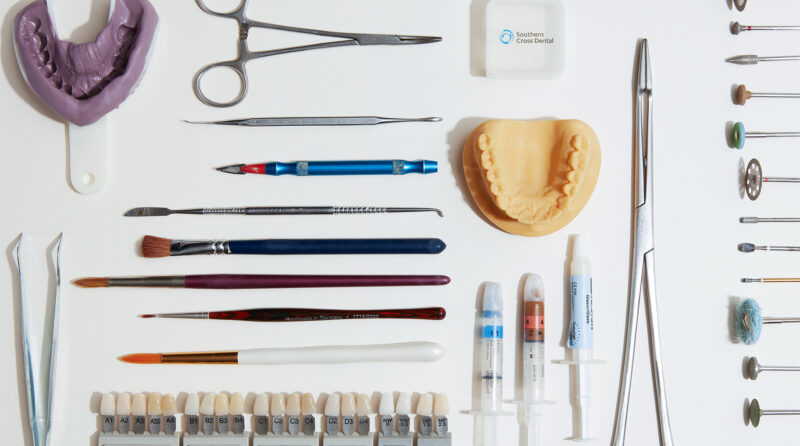
At Southern Cross Dental, we source all our raw materials and components from internally evaluated and approved internationally reputable suppliers: Ivoclar Vivadent, Dental Direkt, Argen and Asiga, to name a few.
If you would like to see data sheets or more information for any of our materials, please get in touch. Our systems ensure identification and traceability from purchase, through manufacture, all the way to sale.
Contact Us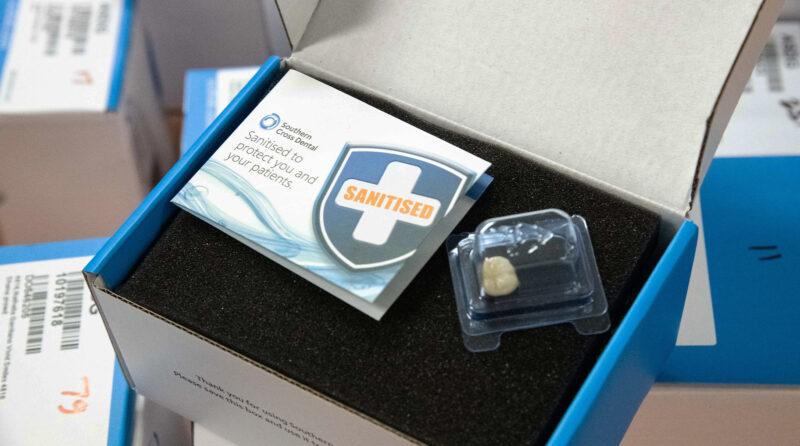
More than ever, it is so important to provide clean and safe practices, and we operate stringent decontamination procedures at Southern Cross Dental. This means that all cases are processed on arrival at our facilities, and returned to you with confidence. Our Standard Range production facilities have received ISO 13485:2016 for medical device manufacturing. All products are sanitized with FDA-approved disinfectants.
Our New Zealand sites operate under an Infection Control Management Plan that is compliant with Australian/New Zealand Standards.
In our Standard Range, product manufacture is also governed by protocols aligned with The Guideline for Disinfection and Sterilization in Healthcare Facilities (Centre for Disease Control, 2008).
Interested to see our infection control practices? Watch here.
Contact us for more information about our best industry practice for safety and compliance.